Lara Marson, a doctoral student in the Department of Applied Microbiology, is investigating which natural derivatives of calcimycin are produced by wild-type and mutant strains under different cultivation conditions.
Microbiology
Piracy among Microbes
Bacteria require iron to grow. If they cannot secure enough on their own, they steal from their neighbors.
Competition rules life in the soil. One billion bacteria live within one gram of dirt, along with fungi. These microbes constantly battle over resources. The fighting is fierce, especially when nutrients are scarce or hard to come by. Professor Julia Bandow and her team are taking a close look at the methods these tiniest of lifeforms use against each other. The researchers’ ultimate goal is to discover new substances that can be used as antibiotics for medical applications. “For example, penicillin is a substance secreted by a fungus to kill competing bacteria, which lyse releasing their cellular components that can then be digested,” says Bandow.
Iron as a limiting factor
Currently, the researchers are focusing on how bacteria find the micro-nutrients they need to grow. These include manganese and iron, which are only required in small amounts but without which growth is impossible. “Iron is an element that is abundant everywhere,” says Bandow. “But it usually occurs in a form that is not readily water-soluble, which makes it difficult for organisms to access.” Iron is one of the limiting factors for bacterial growth. “If it were easy to get, nutrient-rich areas would soon be overgrown with half an inch of bacteria,” the researcher explains.
Not surprisingly, bacteria have developed a range of methods to access iron. The strategies vary in effectiveness. If there is a lot of iron in the soil, an uptake system is all that is needed. If iron is present in an insoluble form or bound to organic material, structurally simple iron-binding molecules—known as siderophores—are secreted to solubilize iron. Citrate would be an example. More complex siderophores such as hydroxamates and catecholates can be used when iron is very low abundant.
Catecholates are the gold standard when it comes to iron availability. “The soil bacterium Bacillus subtilis is a real specialist here,” says Bandow. “It is capable of forming catecholate siderophores that surpass other methods of iron aquisition.” This comes at a high cost to the bacterium, however. Forming these molecules requires a complex synthetic machinery. When the siderophores are released, they form water-soluble complexes with iron that then have to be imported through the cell membrane, which requires energy. And once the complex has been taken up into the bacterium, it has to be broken down enzymatically to release the iron. “Bacillus subtilis only uses this method when iron is scarce,” explains Bandow. “Otherwise, it uses simpler compounds.” This particular bacterium collects iron and stores it within itself so that it has a sufficient supply to support growth.
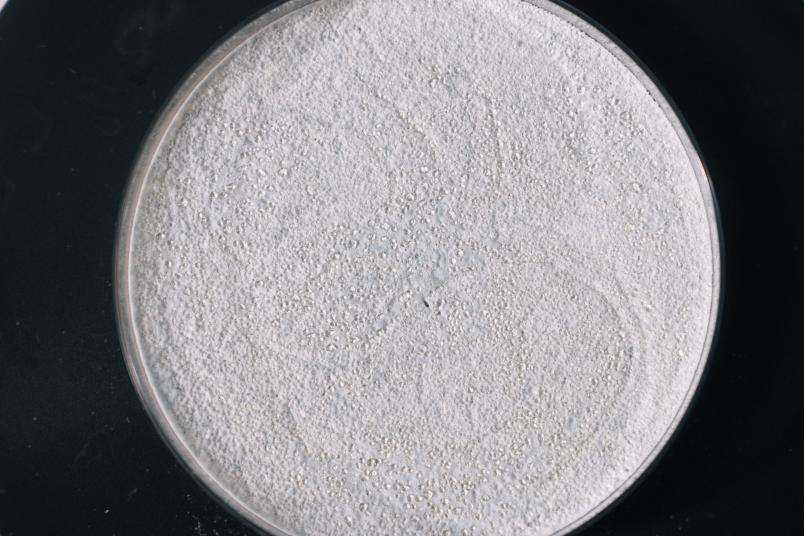
The cultivation of Streptomyces chartreusis on solid medium is used to produce bacterial spores.
One of its competitors, Streptomyces chartreusis, has a different strategy. This soil bacterium is also able to absorb iron from its surroundings. To do this it uses hydroxamate siderophores, which works as long as iron is at least moderately available. If iron is scarce and catecholates are the only solution, Streptomyces relies on a different tactic: It steals from its neighbors. “It has an uptake system for foreign siderophores,” explains Bandow. “This siderophore piracy is quite common among bacteria, and it can also be found in bacteria that colonize the human body.”
Tools for metal theft
Streptomyces chartreusis has another ace up its sleeve. The researchers are investigating ionophores as key substances, especially calcimycin.
In their original state, the carrier ionophores that Streptomyces produces are water-soluble, making it difficult for them to pass through the bacterial lipid membrane. This becomes easier when they bond with a metal atom. Turning the water-soluble side of the molecule toward the metal, the complex exposes a hydrophobic surface. In this form it can pass through the lipophilic layer of the cell membrane passively without expending energy.

Dr. David Podlesainski heads the Mass Spectrometry Core for Life Sciences (MCLS) at the Department of Biology and Biotechnology. He performs elemental analyses that provide information about the metal composition of biological samples.
The researchers isolated the ionophore calcimycin and observed how the substance affects Bacillus subtilis. To do this, they used an ICP mass spectrometer procured by Ruhr University Bochum (ICP stands for “inductively coupled plasma”) that can measure traces of metal. “Just establishing the workflow cost us a lot of time,” says Bandow. “Even the slightest contaminations ruin the measurements. When preparing the samples, you must not wear jewelry or use lotion on face or hands, and of course you have to wear gloves.”
The team learned that upon exposure to calcimycin Bacillus loses iron and manganese. “In exchange, calcium, which is found in high concentrations in the cell’s environment, accumulates within the cell,” Bandow explains. “This carrier ionophore-mediated exchange of matter is driven by diffusion along gradients. You might imagine diffusion processes to be quite slow, but on the size scale of a microbial cell they are actually quite fast.” The loss of micro-nutrients triggers Bacillus to produce more siderophores to rebuild its iron stores.
To learn more about the processes involved, the researchers use modified calcimycin. They discovered that attaching a bromine atom to the ionophore changes its properties. Instead of transporting iron, it now transports copper.
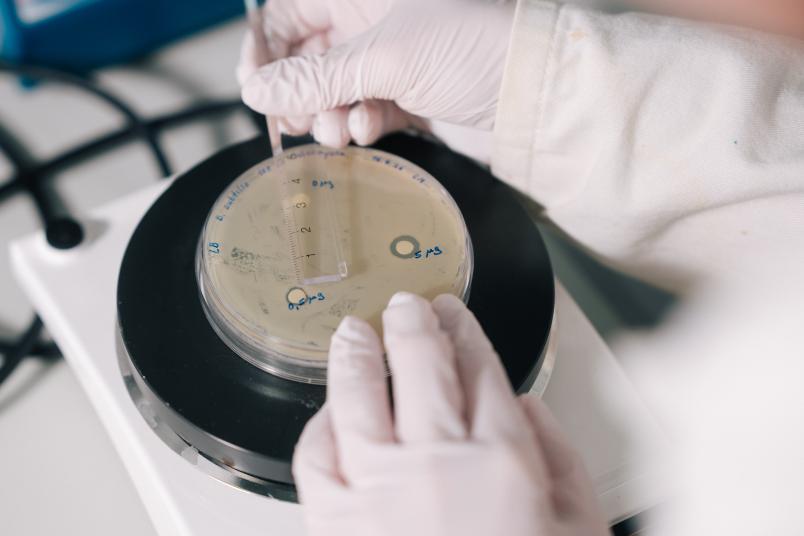
Purified ionophores inhibit the growth of the soil bacterium Bacillus subtilis on nutrient agar. The size of the inhibition zones in the bacterial colony around the calcimycin-soaked filter discs increases as the ionophore concentration rises.
The team purified different ionophores produced by the bacterium. “We were able to show that Streptomyces can realize eight to ten different compounds that only slightly differ chemically,” reports Bandow. “We next want to find out how production of the different variants helps the bacterium to adapt to different environmental conditions.”
Bacteria under suspicion
Based on all they have learned so far, the researchers suspect Streptomyces of systematically robbing Bacillus subtilis. Streptomyces can transport iron out of Bacillus subtilis, take it up with siderophores, thus supplying the iron required to support its own growth.
This would be quite mean of Streptomyces, but might be exploited to our benefit: Ionophores like calcimycin have a strong antibiotic effect.